A radiocarbon determination is a measure of the amount of radiocarbon in a sample. While any organism is alive it continues to incorporate radiocarbon from the atmosphere. Once it has died the amount gradually declines because of radioactive decay.
Measurements of radiocarbon concentration are usually expressed in terms of a notional age, in numbers of years before 1950. For example, the radiocarbon result 1000±25BP indicates that the notional age is 1000 years with a standard uncertainty of 25 years. This notional age is calculated on the simplistic assumption that the amount of radiocarbon in the atmosphere has always been the same. This is not quite the case and so for anything other than a very rough indication of age the measurement must be calibrated.
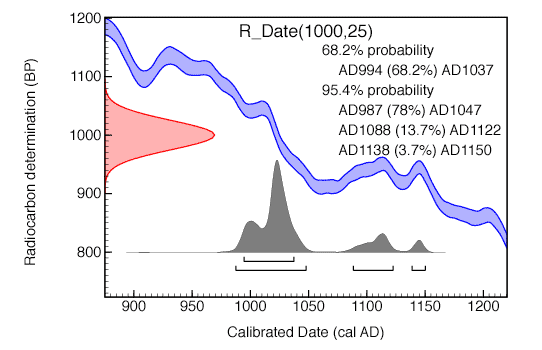
Calibration is performed by comparing the radiocarbon measurements on the sample to those made on material (usually tree rings) of known age. This comparison allows one to determine the possible calendar age of the sample. An example calibration is shown here:

The main elements of this plot are:
The range of possible ages is also shown for two different levels of confidence. We can be 68% sure that the sample dates to between cal AD 994 and cal AD 1037 but there is a reasonable chance (32%) that it is older or younger than this. However we can be 95% certain that it dates to the period cal AD 987 to AD1047, the period cal AD 1088 to cal AD1122 or the period cal AD 1138 to cal AD 1150. The values given within the brackets give the relative likelihood of the individual ranges.
See also Explanation of radiocarbon results from the modern period.